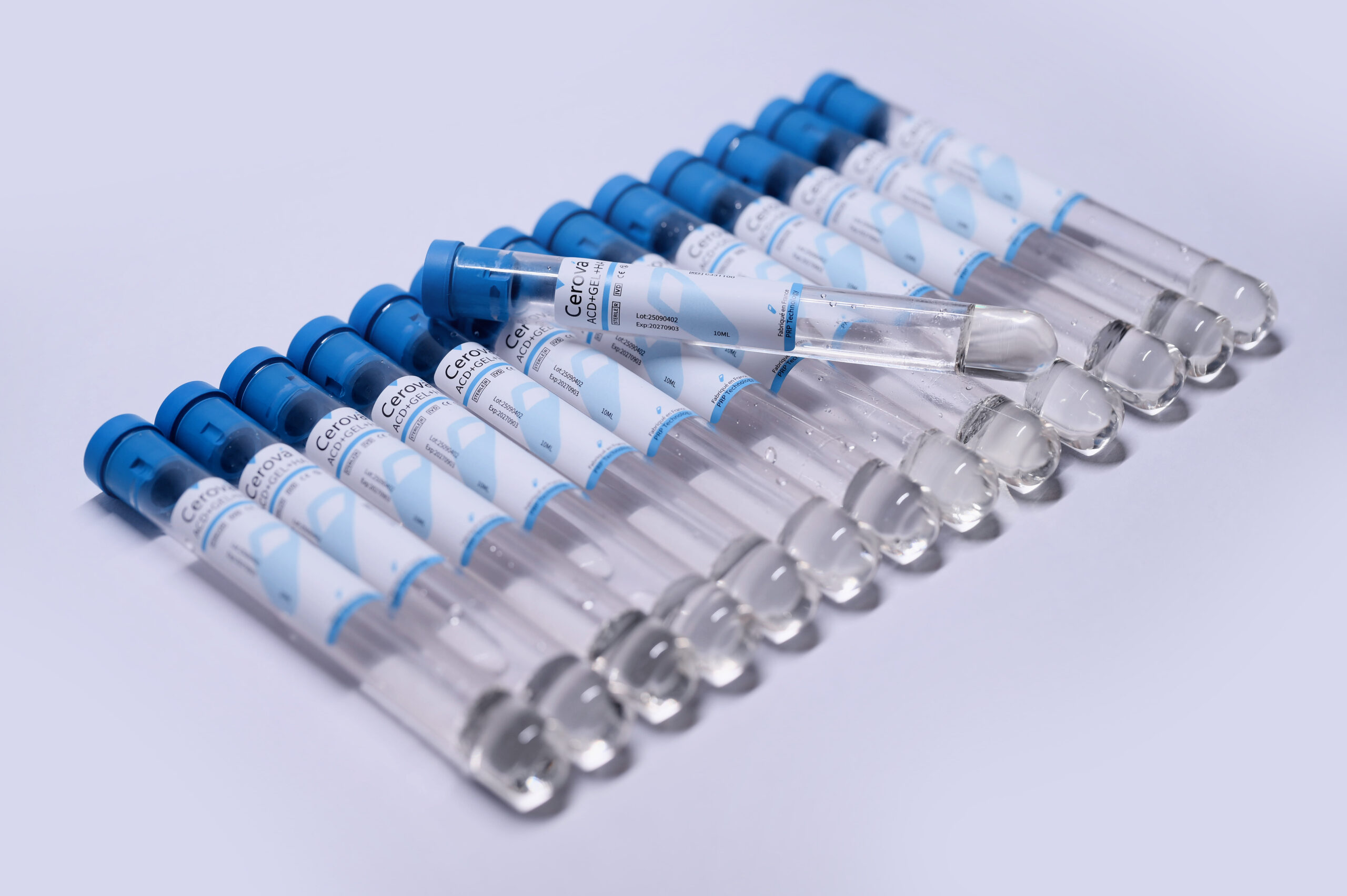
Products
CerovaLabs GFC & PRP Tubes – Precision, Purity, Performance
Designed to elevate your clinic, streamline regenerative treatments, and deliver consistent results for every patient.
" CerovaLabs offers a complete portfolio of PRP and GFC blood collection and preparation tubes, engineered to meet the evolving demands of regenerative medicine. "
CerovaLabs Growth Factor Concentrate (GFC) Kits
Formulation : ACD + Gel, ACD + HA, ACD + Biotin, ACD + Activator
- High-Purity Growth Factor Plasma
Designed to minimize cellular contamination, supporting the stability and functional integrity of growth factors at the time of application. - Preservation-Focused Design
Engineered to protect growth factors from degradation, enabling higher bioavailability and improved regenerative response. - Consistent, Reproducible Outcomes
Reduced variability in plasma quality allows clinics to deliver predictable results across patients and procedures. - Enhanced Patient Confidence
Higher quality regenerative preparations translate into improved healing response and patient satisfaction. - Premium Clinical Positioning
Signals advanced capability and scientific sophistication, elevating the perceived standard of care offered by the clinic.


PRP TUBES (Platelet-Rich Plasma)
Sodium Citrate & Gel PRP Tubes
Designed for effective plasma separation and platelet concentration.
The Cerova PRP Advantage:
- Reliable Plasma Separation
Designed to consistently produce clean plasma suitable for routine regenerative procedures. - Optimized for Daily Clinical Use
Streamlined handling supports fast turnaround without compromising plasma quality. - Predictable Performance
Helps standardize results across treatments, improving practitioner confidence. - Versatile Across Applications
Suitable for dermatology, hair restoration, and musculoskeletal therapies.
QUALITY & SAFETY ACROSS THE RANGE
Every Cerova tube PRP or GFC is manufactured under stringent quality systems and validated through:
GammaSure® Gamma Irradiation Sterilization
Microbial Load Testing (Batch-wise)
Bacterial Endotoxin Testing (BET)
This triple-layer quality assurance ensures biocompatibility, sterility, and clinical reliability across all product lines.